News
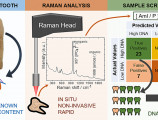
Supported through a Theodore Papazoglou FORTH Synergy Grant (Spectra-Gen: Spectroscopic Screening of Ancient Dental Remains for Optimized Archaeogenetic Analysis), researchers from IESL-Photonics for Heritage Science group, IMBB- Ancient DNA Lab, and ICS, studied a large number of ancient teeth, originating from various excavations and contexts in Greece and across a time span of 8 millennia.
Dr. Giannis Zacharakis, a biophotonics and biomedical imaging researcher and CEO of Kymatonics, was featured in the “Ask Me Anything” interview series by Physics World, where scientists share insights into their careers and the realities of research.
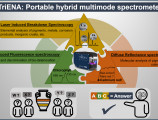
Conceptualised, developed and constructed in the context of the CALLOS project, TRIENA is a compact, portable, field-ready analytical instrument, fusing three spectrochemical techniques: Laser-Induced Breakdown Spectroscopy, LED-Induced Fluorescence, and Diffuse Reflectance spectroscopy.